About Lipella Pharmaceuticals, Inc.
Lipella Pharmaceuticals is a clinical-stage biotechnology company developing and commercializing treatments for serious diseases. Our proprietary drug delivery technology has potential applications in addressing diseases of the mucosal tissue including the bladder, urethra, oral cavity, esophagus and colon. We see strong potential for our technology in partnerships and commercial licensing agreements.
Lipella maintains a sterile manufacturing facility in Pittsburgh, PA for the production of clinical supplies and research products.
Click the button above to view Lipella Corporate Presentation
July 2024

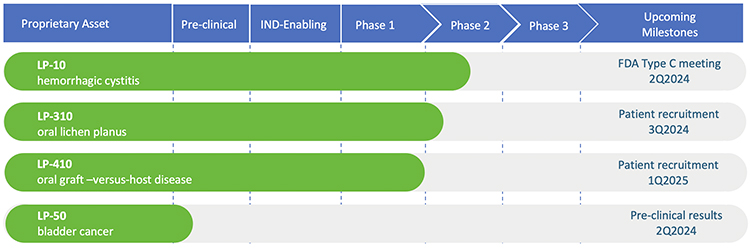
We recently completed a phase 2A multi-center dose-escalation clinical trial for our lead product, LP-10. Results are published in the peer-reviewed journal, International Urology and Nephrology.
Our pipeline asset, LP-310 , was approved by the FDA to begin phase 2A multi-center, dose-escalation clinical trial to assess the safety and efficacy of LP-310 in patients with symptomatic oral lichen planus (OLP).
We recently received Orphan Drug Designation from the FDA for our drug candidate, LP-410, which has been designed for the treatment of oral Graft-versus-Host Disease (GVHD). Oral GVHD is a rare but serious oral mucosal disease that currently has no approved therapy. Lipella’s proprietary oral rinse formulation of liposomal tacrolimus has the potential to effectively treat oral GVHD, while minimizing systemic toxicity.
We maintain a pipeline of additional products under development.

Reach Out